Aldehyde ir spectrum12/10/2023 This argument however, fails to explain why the carboxylic acid has a stretching frequency below that of the ketone, since, as was already said, they both have the oxygen adjacent to the carbonyl. We can explain the ester being stronger by considering the effect of the electronegative oxygen withdrawing electron density: Therefore using your data we can say that the ester is stronger than the ketone whilst the acid is weaker. The frequency range for acid chlorides is higher than that for aldehydes due to the presence of the electron-withdrawing chlorine atom. 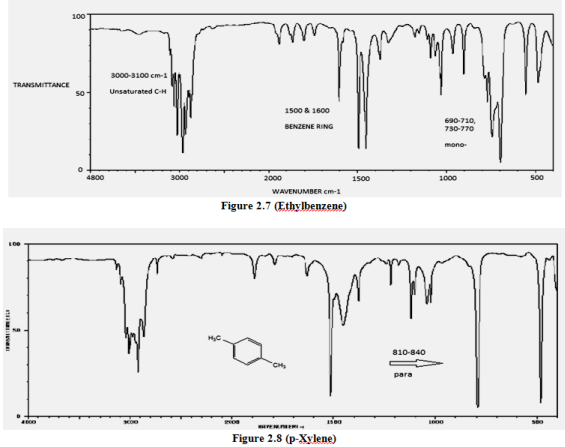
In your statement, you may have gotten a little confused, remember that the stretching frequency increases with bond strength. This must be as a result of the fact that in the ester inductive withdrawal is more effective than resonance and the converse is true in the acid. That said, the difference between the frequency range of an acid vs an ester is an interesting one, as on the surface of it they are electronically quite similar (in both cases the carbonyl is next to an electronegative This would seem to suggest that the acid decreases the carbonyl bond strength and the ester weakens it. Stretching frequencies of common carbonyl groups (Taken from Introduction to Spectroscopy, Pavia and Lampman. The mid-IR spectra presented here for amorphous. Ketones and acids, for example, cover a very similar portion of the spectrum. The overall agreement between our IR spectra in Figs 2 5, and our peak positions of Tables 1 and 2, and similar results from earlier workers is quite good, even allowing for differences in the ways that the ice samples were prepared or the methods for recording IR spectra. 
I've provided the table below, from which you can see that actually there is a great deal of overlap depending upon the actual 'environment' of the functional group in question. First things first, it's important to realise that functional groups may have IR frequencies over quite a broad range.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
