Emulsion examples chemistry12/12/2023 In four glasses or test tubes place 2.5ml vinegar and 2.Emulsions are thicker than either the water or of fat/oil they contain, which is a useful property for some foods. For example, aerosol (Examples Clouds, Fog etc.) is also a colloidal solution in which the dispersion medium is gas and the dispersed phase is. Aerosol contains small particles of liquid or solid dispersed in a gas. Why is this mixture called a temporary emulsion Where else have you seen an emulsion One example is an oil spill in the ocean. The phrase 'colloidal solution' is incorrect. Also you will learn about the difference between solution s. The mixture may be termed a colloid or a colloidal suspension. In this animated lecture, I will teach you about solution, suspension, colloid and true solution. The insoluble particles in the mixture are microscopic, with particle sizes between nanometers. Foam is formed when many gas particles are trapped in a liquid or solid. A colloid is a type of homogeneous mixture in which the dispersed particles do not settle out. Sol is a colloidal suspension with solid particles in a liquid. In colloidal solution, both dispersed phase and dispersion medium don’t need to always be liquids. The types of colloids includes sol, emulsion, foam, and aerosol. By vigorously mixing the emulsifier with the water and fat/oil, a stable emulsion can be made.Ĭommonly used emulsifiers include egg yolk, or mustard. Although emulsion is a type of colloid, it does not mean that all colloids are emulsions only. The hydrophilic end of the emulsifier molecule is attracted to the water and the hydrophobic end is attracted to the fat/oil. These help to form and stabilise the emulsions, preventing or slowing the water and fat/oil from separating.Įmulsifier molecules work by having a hydrophilic end (water-loving) and hydrophobic end (water-hating). In smoke, for examples, solid particles from combustion are suspended in a gas. 
However, the particles in a colloid are smaller than those found in a suspension. These particles are larger than molecules, distinguishing a colloid from a solution. 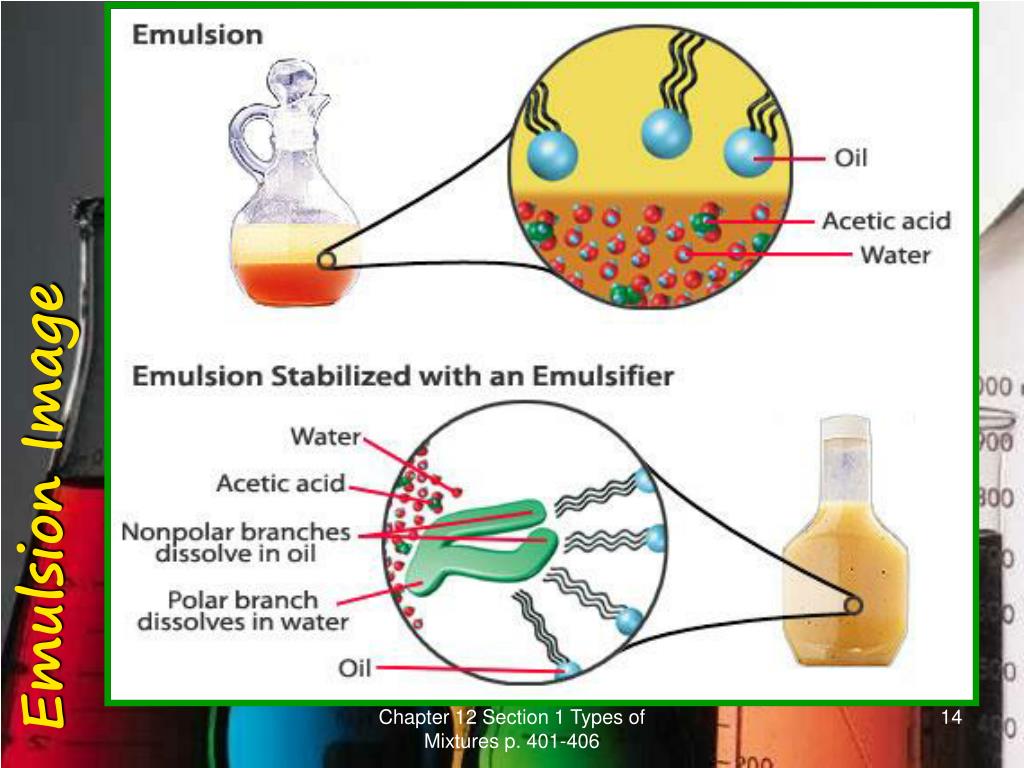
To prevent the mixture from separating substances called emulsifiers can be added. The colloid particles are solids or liquids that are suspended in the medium. However the mixture is unstable and if you left it for a while it would soon separate out into water and oil layers again. If you shake the oil and water together then the oil breaks up into tiny droplets and becomes distributed in the water forming a mixture. In this type of emulsion the dispersed phase. If you add a drop or two of oil to water you can see that it does not dissolve or combine with the water: the oil floats on the water. Milk is an example of this type of emulsion in which liquid fat is dispesed in water.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
